Extracorporeal Membrane Oxygenation, or ECMO for short, is an advanced therapy that is sometimes used to treat people with life-threatening heart and lung failure, to do the work of the heart and lungs when a patient’s own organs are too sick or weak to work on their own. It is effectively a modified heart-lung bypass machine—a machine that takes over heart and lung function (meaning it adds oxygen to and removes carbon dioxide from a patient’s blood supply).
But unlike a heart-lung bypass machine, which is designed for short-term use (during heart surgery, for instance), ECMO machines provide long-term heart and lung support over a period of hours, days, or even weeks to give a patient’s heart and lungs time to heal and regain function. It provides a kind of bridge, a temporarily replacement that keeps the functions of the heart and lungs going while doctors treat the underlying problem.
ECMO can be used for patients of all ages, from infants to adults. It can help patients with a range of severe heart and lung conditions, from cardiac arrest to respiratory failure. But in most cases, ECMO therapy is used only when all other conventional treatments have failed to resolve the underlying heart or lung disorders.
 |
| ECMO Machine |
 | |||||||
| The complete ECMO Machine | c |
Connecting To ECMO
Connecting a patient to the ECMO machine requires surgery. The ECMO machine connects to a patient through plastic tubes called cannulas. After giving the patient an anticoagulant, a medication that prevents blood from clotting, the doctor inserts cannulas into large arteries and veins located in the chest, neck, or legs. Once connected, the ECMO machine draws blood from the patient, which it then passes through the cannulas and into an artificial lung that infuses the blood with oxygen and removes carbon dioxide. The ECMO machine then warms this treated blood to body temperature and pumps it back into the patient. In cases where a patient’s heart cannot circulate blood on its own, a mechanical pump takes over the heart’s role and pumps blood through the patient’s circulatory system.
ECMO therapy is frequently a treatment of last resort. It is recommended when other treatments have failed to resolve the underlying problem, but there is still a possibility of recovery.
The doctors continue to administer sedatives and pain medications after surgery to keep patients as comfortable and pain-free as possible, perform routine chest X-rays, run regular blood tests to assess oxygen and carbon dioxide levels that allow doctors to evaluate and track the patient’s lung and heart health and to check for possible infections.
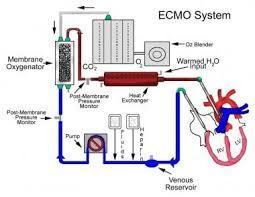 |
| Basic setting up of the ECMO |
 |
| Connecting to a patient |
Applications
While there is no fixed list of conditions for which ECMO is used, doctors may recommend its use in the following situations:
- Respiratory failure (when the lungs fail to maintain adequate oxygen levels or remove enough carbon dioxide from the blood)
- Heart transplantation
- Lung transplantation
- Cardiac arrest (when the heart fails to pump blood effectively)
- Cardiogenic shock (when the ventricles of the heart do not function properly, resulting in insufficient blood flow)
- Pulmonary embolism (when an artery in the lungs is blocked)
- Birth defects of the heart
- Acute Respiratory Distress Syndrome, ARDS (a type of respiratory failure that prevents adequate oxygen from getting to the lungs and blood)
Why Is ECMO used in some COVID-19 Patients?
In general, doctors aim to take patients off ECMO therapy as quickly as possible. Because it is used for patients with a range of conditions, each with its own recovery timetable, the length of time someone is on ECMO therapy can vary greatly. Some patients need it for only a few hours while others may require days or weeks of ECMO support.
Once the patient reaches a point at which the ECMO machine is no longer necessary, the ECMO team will begin the process of weaning in which they steadily decrease the patient’s blood flow through the machine. Over several hours, they track the patient’s response to this reduction in ECMO support. If the patient responds well, and the ECMO team concludes that discontinuation of ECMO is safe, a surgeon will remove the cannulas.
After coming off ECMO, a patient might require a ventilator to provide breathing support. As soon as the patient is able to breathe without assistance, doctors will remove the ventilator. But patients may still need to stay in the hospital for days or weeks, at least until vital signs are stable. Many will also need physical therapy to help regain muscle strength, as well as speech therapy to aid recovery after long-term use of a ventilator’s breathing tube.
Ref: Youtube.com: "I almost died" by CNA
What are the risks associated with ECMO?
- Bleeding: Bleeding affects up to 50% of ECMO patients and, in some cases, can be life-threatening. ECMO therapy requires the use of anticoagulants, medications that thin the blood (and prevent the formation of clots) to keep the blood flowing through the body and ECMO machine. But these medications also increase the likelihood of excess bleeding. As a result, hemorrhaging may occur in a number of places, including the lungs, stomach, mouth, nose, and brain. Doctors watch for signs of bleeding; medication or surgery may be necessary to stop it.
- Kidney Failure: In some cases, patients on ECMO do not circulate enough blood to their kidneys, resulting in kidney failure and the potential need for dialysis, a machine that does some of the work normally done by kidneys. Kidney function usually returns after the patient is removed from the ECMO machine, though sometimes the kidneys do not recover, and the patient will require lifelong dialysis treatment.
- Bacterial Infection: Because cannulas are inserted into veins and arteries in ECMO therapy, bacteria can have direct access to the patient’s blood stream. If unchecked, infections may result, such as bacterial pneumonia. If infection is suspected, patients are treated with a course of antibiotics and cannulas may be replaced.
- Stroke: In rare cases, ECMO patients develop small blood clots that can reduce the flow of blood to the brain. This raises risk for stroke.
- Pulmonary Embolism: Though it is rare, some patients on ECMO develop a blood clot that blocks blood flow in the lungs. Pulmonary embolism can cause permanent damage to the lungs, as well as damage to other organs as the lungs may be unable to provide enough oxygen to the body.
Reference: www.yalemedicine.org/conditions/ecmo





